CAR-T Therapy Market by 2031 – Growth Drivers, Opportunities, and Industry Outlook
The CAR-T Therapy Market is rapidly evolving as immunotherapy technologies reshape the treatment landscape for cancer worldwide. Chimeric Antigen Receptor T-cell (CAR-T) therapy is an innovative treatment approach in which a patient’s immune cells are genetically engineered to recognize and destroy cancer cells. This personalized therapy has demonstrated exceptional clinical outcomes in treating blood cancers such as leukemia, lymphoma, and multiple myeloma. As healthcare systems increasingly shift toward precision medicine and targeted therapies, CAR-T therapy is emerging as one of the most transformative breakthroughs in oncology.
The CAR-T Therapy Market is expected to reach US$ 31.52 billion by 2031, expanding at a strong CAGR of 34.8% during 2025–2031. The increasing incidence of cancer, rising clinical adoption of immunotherapy, and continuous advancements in cell engineering technologies are key factors supporting this growth trajectory. The market report scope includes analysis based on targeted antigen, therapeutic application, and regional landscape, providing stakeholders with a comprehensive understanding of emerging market opportunities and strategic developments.
Download Sample PDF: https://www.theinsightpartners.com/sample/TIPRE00003731
Market Drivers Fueling the Growth of the CAR-T Therapy Market
Rising Global Cancer Incidence
One of the most significant drivers of the CAR-T therapy market is the increasing global burden of cancer. Hematological malignancies such as acute lymphocytic leukemia and diffuse large B-cell lymphoma continue to affect millions of people worldwide. Conventional treatment options such as chemotherapy and radiation therapy often fail to provide long-term remission for patients with relapsed or refractory cancers.
CAR-T therapy offers a targeted and highly personalized treatment approach that reprograms immune cells to identify and eliminate cancer cells more effectively. The increasing demand for advanced treatment options for complex cancers is encouraging healthcare providers to adopt CAR-T therapies, thereby accelerating market growth.
Growing Regulatory Approvals for CAR-T Therapies
The expanding number of regulatory approvals for CAR-T therapies is another major factor driving market expansion. Regulatory agencies across North America and Europe are increasingly approving CAR-T products as clinical trials demonstrate their safety and effectiveness.
These approvals have accelerated the commercialization of CAR-T therapies and strengthened confidence among healthcare professionals and investors. The development of regulatory frameworks specifically designed for advanced therapy medicinal products is also encouraging biotechnology companies to accelerate their research and product development initiatives.
Technological Advancements in Gene and Cell Engineering
Rapid technological progress in gene editing and cell engineering is transforming the CAR-T therapy landscape. Advances in viral vector technology, genome editing tools, and cell manufacturing platforms are improving the safety, efficiency, and scalability of CAR-T treatments.
Researchers are developing next-generation CAR constructs that enhance antigen targeting while reducing treatment toxicity. Dual-target CAR-T therapies and advanced genetic modifications are also being explored to overcome tumor resistance and improve clinical outcomes. These innovations are expected to significantly expand the therapeutic potential of CAR-T therapy in the coming years.
Emerging Opportunities in the CAR-T Therapy Market
Expansion into Solid Tumor Treatment
While CAR-T therapy has primarily been used to treat blood cancers, researchers are increasingly focusing on developing CAR-T therapies for solid tumors such as breast cancer, pancreatic cancer, and colorectal cancer. The successful development of CAR-T treatments for solid tumors would significantly expand the addressable market and unlock new growth opportunities for biotechnology companies.
Advances in tumor microenvironment research and antigen discovery are helping scientists overcome challenges associated with treating solid tumors using CAR-T therapy.
Development of Off-the-Shelf CAR-T Therapies
Another promising opportunity in the CAR-T therapy market is the development of allogeneic or “off-the-shelf” CAR-T therapies. Unlike traditional CAR-T treatments that require patient-specific cell engineering, off-the-shelf therapies utilize donor-derived cells that can be manufactured in advance.
This innovation has the potential to reduce production costs, shorten treatment timelines, and improve accessibility for patients. As biotechnology companies invest heavily in allogeneic CAR-T research, these therapies are expected to reshape the future of the market.
Market Segmentation Insights
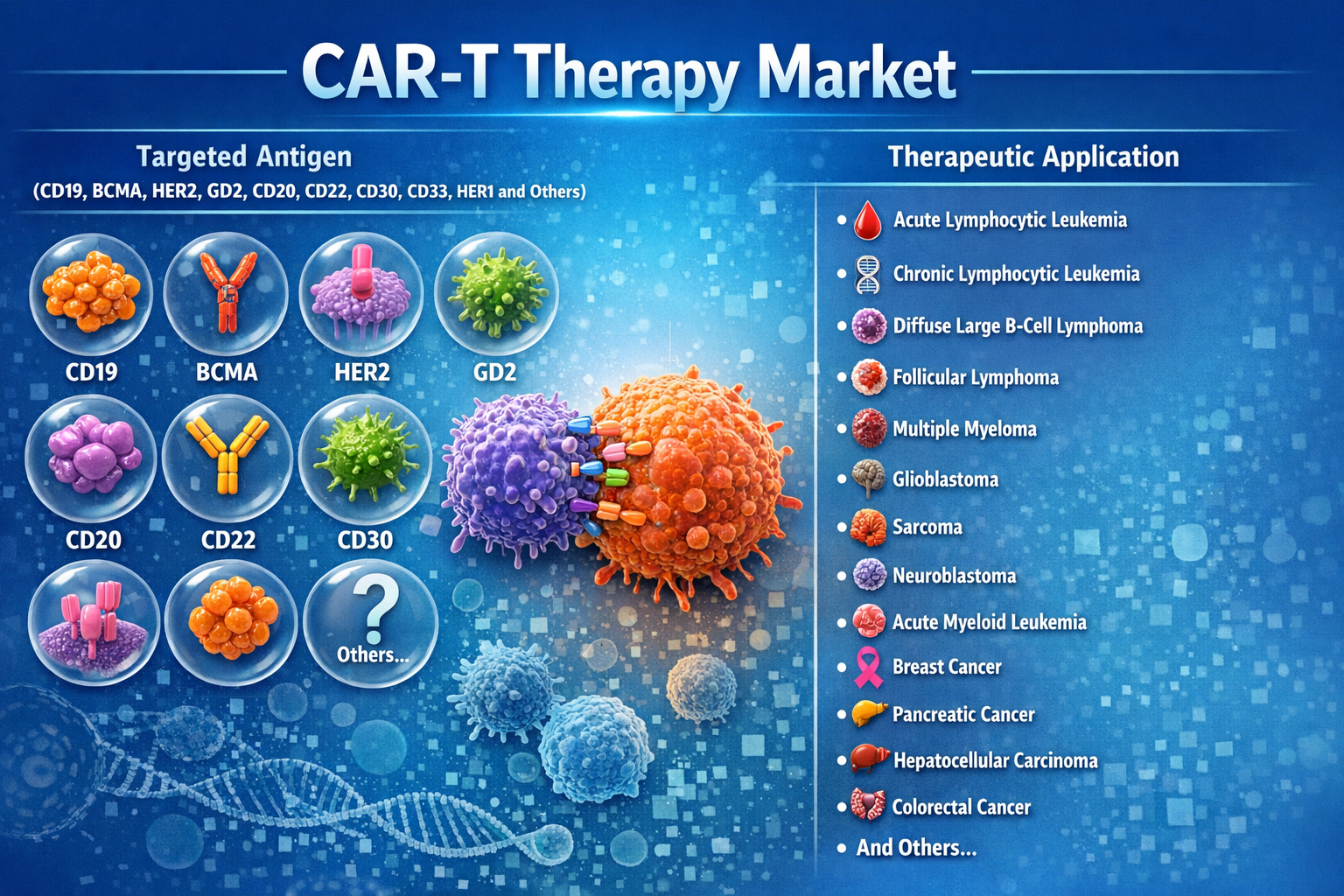
The CAR-T therapy market is segmented based on targeted antigen, therapeutic application, and geography.
By targeted antigen, the market includes CD19, BCMA, HER2, GD2, CD20, CD22, CD30, CD33, HER1, and others. Among these, CD19-targeted CAR-T therapies dominate the market due to their strong clinical performance in treating B-cell malignancies.
By therapeutic application, CAR-T therapies are widely used for conditions such as acute lymphocytic leukemia, chronic lymphocytic leukemia, diffuse large B-cell lymphoma, follicular lymphoma, and multiple myeloma. Research is also expanding into other cancers including glioblastoma, neuroblastoma, pancreatic cancer, hepatocellular carcinoma, and colorectal cancer.
Geographically, North America currently dominates the market, supported by strong biotechnology infrastructure, early adoption of advanced therapies, and significant research investments.
Key Companies in the CAR-T Therapy Market
The competitive landscape of the CAR-T therapy market includes several global biotechnology and pharmaceutical companies actively investing in product development and clinical research. Key market players include:
- Novartis International AG
- Kite Pharma, Inc. (Gilead Sciences, Inc.)
- Juno Therapeutics (Celgene Corporation)
- Bluebird Bio, Inc.
- Sorrento Therapeutics Inc.
- Mustang Bio, Inc.
- Aurora Biopharma Inc.
- Legend Biotech (Genscript Biotech Corporation)
- Pfizer, Inc.
These companies are focusing on expanding their CAR-T therapy pipelines, improving manufacturing capabilities, and forming strategic partnerships to strengthen their global market presence.
Future Outlook of the CAR-T Therapy Market
The long-term outlook for the CAR-T therapy market remains highly promising as innovations in biotechnology continue to accelerate the development of advanced cancer treatments. Industry studies suggest that the market could experience exponential growth in the coming decade as personalized medicine becomes more widely adopted. Some projections indicate the global CAR-T therapy market could surpass US$ 146 billion by 2034, driven by strong research pipelines and increasing demand for targeted immunotherapies.
Advancements in gene editing technologies, improved manufacturing processes, and increasing global investments in biotechnology will continue to expand the clinical potential of CAR-T therapies. As the therapy evolves and becomes more accessible, it is expected to play a central role in the future of precision oncology and cancer treatment.
About Us
The Insight Partners is a leading global market research and consulting firm that provides actionable insights and strategic intelligence across multiple industries including healthcare, technology, energy, and manufacturing. Our research reports deliver comprehensive market analysis, competitive benchmarking, and future growth forecasts to help organizations make informed business decisions.
Recent Report
Cardiometabolic Diseases Market Share, Size and Future Opportunities 2034





